 Pakistan has had a questionable engagement with the COVID–19 pandemic with analysts terming the state’s response as complacent, supplemented by a refusal to follow social–distancing guidance by congregating for prayers and living in densely populated regions with shared communal spaces. With the lack of sufficient healthcare infrastructure, Pakistan should be teetering on the edge of potential disaster—but it has not done so yet. Of Pakistan’s 219 million population there have been 4489 cases—of which 65 have died. These are much better numbers than Italy (18279 deaths), Spain (15238 deaths), France (12210 deaths) and UK (7978 deaths)—all of which have much lower populations and population densities than Pakistan. This corresponds to a scientific debate—on the prophylactic effects of anti–malarial drug hydroxychloroquine found in malaria effected regions like South Asia and Africa—one most vociferously fought in the United States.
Pakistan has had a questionable engagement with the COVID–19 pandemic with analysts terming the state’s response as complacent, supplemented by a refusal to follow social–distancing guidance by congregating for prayers and living in densely populated regions with shared communal spaces. With the lack of sufficient healthcare infrastructure, Pakistan should be teetering on the edge of potential disaster—but it has not done so yet. Of Pakistan’s 219 million population there have been 4489 cases—of which 65 have died. These are much better numbers than Italy (18279 deaths), Spain (15238 deaths), France (12210 deaths) and UK (7978 deaths)—all of which have much lower populations and population densities than Pakistan. This corresponds to a scientific debate—on the prophylactic effects of anti–malarial drug hydroxychloroquine found in malaria effected regions like South Asia and Africa—one most vociferously fought in the United States.
On March 21st, US President Donald Trump tweeted, “Hydroxychloroquine & Azithromycin, taken together, have a real chance to be one of the biggest game changers in the history of medicine.” On Sunday March 29th, the United States Food and Drug Administration (FDA) issued an emergency use authorization for these “decades–old malaria drugs”, to be “donated to the Strategic National Stockpile”. Moreover, they were green–lit to be “prescribed by doctors to hospitalized teen and adult patients with COVID-19” as stated by the Department of Health and Human Services (HHS) with their announcement of German pharmaceutical giant Sandoz donating 30 million doses of hydroxychloroquine to the national stockpile. All this happened in the span of two weeks—with a March 13th tweet by cryptocurrency investor James Todaro and New York City lawyer Gregory Rigano sharing a Google Doc link to a paper discussing chloroquine trials in China and France showing a prophylactic potential against COVID–19. With SpaceX CEO Elon Musk and US right–wing media outlets like Breitbart and The Blaze touting its advantages, US President Donald Trump entered the stage. On March 23rd, the US FDA lifted import sanctions on Ipca Laboratories ltd.—a Mumbai–based pharmaceutical company—in order to increase supplies of chloroquine and hydroxychloroquine, allowing the supply of tablets and raw materials. And this Sunday, the drug use was put into practice.
Medical experts from Yale’s Interdisciplinary Center for Bioethics and University of Washington have criticised the President’s remarks as irresponsible, advising against spearheading mass–consumption efforts before following the due process of clinical trials. Even in miniscule doses, chloroquine is still notably toxic and has seen patients being hospitalised in Wuhan and Nigeria. It poses a risk of heart damage and potential congenital birth defects, which could be a rationale for many countries abstaining from its subscription. Yet a large amount of research is indicating the contrary, highlighting benefits of the drug tested on in vitro cases. A joint paper in February 2020 by Wuhan Institute of Virology and Beijing Institute of Pharmacology and Toxicology referred to chloroquine as a “potential broad–spectrum antiviral drug” and one which is “highly effective in the control of 2019–nCoV infection in vitro”. A similar assertion by a joint–research from South London and Maudsley NHS Trust and Kings College London contended a similar case through the use of cartographic comparison of malaria and COVID regions, as replicated in Figure 1. According to the study, the geographical distribution exhibits a negative correlation between the prevalence of anti–malarial prophylaxis and the COVID–19 infection, which they concluded was due to immunity provided through the consumption of hydroxychloroquine.
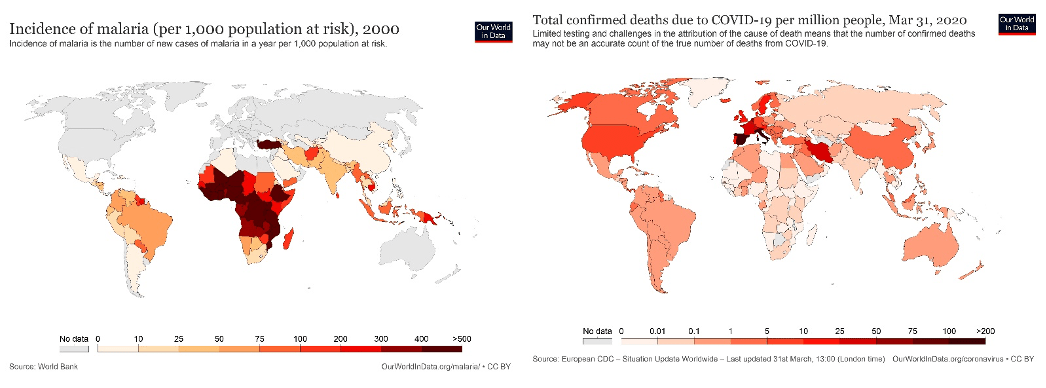 Data acquired: Malaria Map https://ourworldindata.org/malaria Coronavirus Map https://ourworldindata.org/coronavirus
Data acquired: Malaria Map https://ourworldindata.org/malaria Coronavirus Map https://ourworldindata.org/coronavirus
The researchers drew reference from a study published by Vincent et al. (2005) in Virology Journal, who studied the antiviral effects of chloroquine on SARS–CoV in an in vitro primate cells study. This investigation was further supplemented by more recent research by Yao et al. (2020), Wang et al. (2020) and Touret & De Lamballerie (2020), all concluding a negative correlation between the prevalence of hydroxychloroquine–induced anti–malarial prophylaxis and COVID–19 infection. As a step in the direction of scientific research, the University of Oxford began clinical trials on Hydroxychloroquine Prevention of Coronavirus Disease in March which are now progressing to human trials. Off–label usage—using an approved drug for unapproved indications based on scientific evidence—is the current case for Hydroxychloroquine. In the absence of a definitive cure, testing the curative potential of this investigational experiment treatment merits significance. Regions with large incidents of malaria and anti–malarial drug use—South Asia and Africa—can thus serve as perfect grounds for herd immunity resistance testing.
Whether one may argue in favour of geographic factors like humidity or exposure, it is fair to say that the discussion on hydroxychloroquine is contested and inconclusive. The abovementioned discussion, thus, warrants attention and serves as a suitable starting point for conducting experimental treatment—in countries like Pakistan—to supplement extant clinical trials. The capital city of Islamabad—and its legislated territory—can serve as Pakistan’s best starting point for experimental treatment. With a joint population of over 2 million residents, the Islamabad Capital Territory (ICT) includes the cities of Islamabad and Rawalpindi, exhibiting the lowest population density in Pakistan’s regional territories and hence best suited for containment measures. Of the confirmed 4489 cases in Pakistan, Islamabad has seen 102 which is 2.3% of the total, the lowest in comparative metrics. Moreover, 4 of the top 10 hospitals in addition to medical facilities of the Armed Forces are in the country are in the ICT region, with substantial resources to facilitate research into experimental treatment. The dire state of Pakistan’s healthcare infrastructure and budgetary restraints notwithstanding, Islamabad’s current facilities can sufficiently conduct hydroxychloroquine trials on the affected patients and allow the PTI government to play a role in medically–combatting this global pandemic.
![]()




Be the first to comment